News
Key molecule in scleroderma onset identified
Scleroderma is an invalidating autoimmune disease which is difficult to diagnose and still lacks effective treatments. A study published today in Science Translational Medicine shows for the first time ever the key role of a specific molecule as a coordinator of the attack by the immune system against tissues. The molecule, called HMGB1, is released into the bloodstream by platelets and acts in a way that modifies metabolism and function of certain immune system cells, leading to chronic damage of small vessels and to fibrosis. If confirmed by further studies, the mechanism discovered might become the first therapeutic target of the disease. The discovery was made by a group of researchers of Ospedale San Raffaele and Università Vita-Salute San Raffaele guided by Angelo Manfredi, head of the Autoimmunity and vascular inflammation Unit. The study has been supported by the Italian association Associazione Italiana Lotta alla Sclerodermia (AILS).
 Scleroderma is also called systemic sclerosis and causes inflammation and occlusion of small blood vessels as well as progressive thickening (fibrosis) of the skin and internal connective tissues. Fibrosis is caused by the continuous attack made by the immune system against tissues, which leads to chronic and dysfunctional cicatrization. According to the organs affected, fibrosis may cause severe complications (to lungs, etc.). The mechanism underlying such an abnormal immune activation has hardly been understood up to now, preventing scientists from developing effective treatments.
Scleroderma is also called systemic sclerosis and causes inflammation and occlusion of small blood vessels as well as progressive thickening (fibrosis) of the skin and internal connective tissues. Fibrosis is caused by the continuous attack made by the immune system against tissues, which leads to chronic and dysfunctional cicatrization. According to the organs affected, fibrosis may cause severe complications (to lungs, etc.). The mechanism underlying such an abnormal immune activation has hardly been understood up to now, preventing scientists from developing effective treatments.

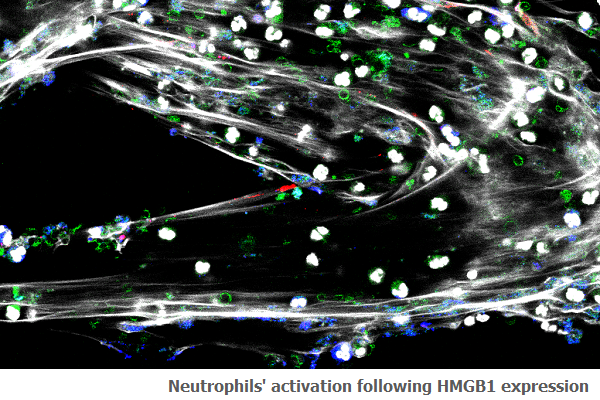
Things might change thanks to this study. The research team identified the role of a single molecule – HMGB1 – as a trigger for specific mechanisms underlying scleroderma. Scientists discovered that the the blood of people with scleroderma is rich in microparticles expressing HMGB1 on their surface. Such micro-particles are introduced into the bloodstream by platelets, which as a consequence contain less HMGB1 molecules than healthy people’s platelets. “We showed both in vitro and in an animal model of the disease that the presence of micro-particles expressing HMGB1 is enough to activate the immune system and in particular neutrophils” says Norma Maugeri, the study’s first author. Neutrophils start releasing their nucleus’ content, included their DNA, and that leads to inflammation and damage of the surrounding tissues. Neutrophils activated by HMGB1, moreover, become ‘immortal’, i.e. reject suppression signals that normally are activated in such abnormal conditions.
While it damages tissues, HMGB1 also promotes their regeneration. Uncontrolled regeneration, though, may lead to fibrosis. The fact that HMGB1 regulates both inflammation and regeneration has already been shown by another team of researchers of Ospedale San Raffaele, guided by Marco Emilio Bianchi, among the authors of the study in Science Translational Medicine.
“Obviously the results we obtained must be further confirmed and expanded, yet we believe that an excessive amount of HMGB1 outside the cells might be the main cause of damage to vessels and fibrosis of connective tissues” says Angelo Manfredi. If it is so, HMGB1 might become a therapeutic target to fight scleroderma – interfering with platelets that release the protein in the bloodstream or eliminating it might hinder the evolution of the disease.
Maugeri N, Capobianco A, Rovere-Querini P, Ramirez GA, Tombetti E, Della Valle P, Monno A, D’Alberti V, Gasparri AM, Franchini S, D’Angelo A, Bianchi ME and Manfredi AA. Platelet microparticles sustain autophagy-associated activation of neutrophils in systemic sclerosis. Science Translational Medicine Vol. 10, Issue 451, 25 July 2018
